Cardiac Arrest Program
Phase 2 Results
In a Phase 2 Clinical Study of 110 patients, published in JAMA, XENEX demonstrated significantly less brain damage than standard-of-care (SOC) alone in OHCA patients. The primary endpoint was reduction in cerebral white matter damage using MRI as a biomarker. Analysis of the global fractional anisotropy (GFA) measured the differences in diffusion of water in white matter tracts of brain to assess damage. The results were 42% less white matter damage, P=0.006. The primary endpoint of brain damage was the leading predictor of mortality at 6 months.
Although the study was not powered for significant secondary endpoints, it surprisingly demonstrated a strong trend toward reduced mortality, with a 20% relative reduction, p=0.053, in the intention-to-treat population (ITT). In a post-hoc analysis of ~90% of the ITT population, it was found that there was a 40% relative reduction in the mortality rate, p=0.05.
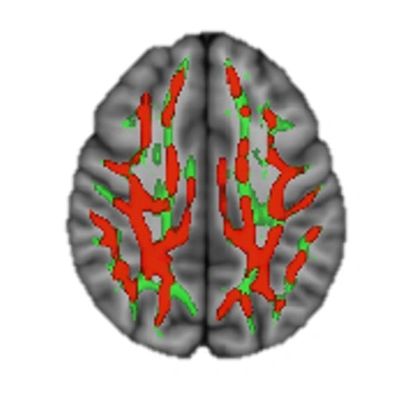
Comparison of Superior Cognitive Region of Brains
Red – significantly less damage in Xenon+SOC group
Cardiac Arrest Program
Phase 3 Trial
The results of the Phase 2 study informed the Phase 3 trial design and served as the data set for regulatory approval for the trial. Based on these robust findings and the significant unmet need, the FDA granted XENEX Fast Track designation and Special Protocol Assessment (SPA) agreement with only a single pivotal registration trial for approval. This single pivotal trial mitigates the risk typically associated with conducting two separate pivotal randomized controlled trials (RCTs) for approval. By enriching the trial with the post‑hoc–defined responder population, we further decrease development and regulatory risk.
XENEX benefits from orphan‑drug designation in post–cardiac arrest, a major regulatory advantage that delivers close to a decade of market exclusivity after approval.

Severe Traumatic Brain Injury
Phase 2 Trial
Traumatic brain injury triggers the same destructive secondary injury cascade seen after cardiac arrest — excitotoxicity, inflammation, impaired cerebral blood flow, mitochondrial dysfunction, and progressive neuronal loss. Despite decades of research, no neuroprotective pharmacotherapies exist, leaving clinicians with only supportive care while patients face poor survival and lifelong cognitive and functional impairment. Preclinical studies across rodent and large‑animal TBI models demonstrate that xenon can interrupt multiple early drivers of secondary injury through its broad, multi‑modal mechanisms. XENEX delivers this neuroprotection rapidly and safely through a practical, closed‑circuit system designed for real‑world emergency and critical care environments, addressing the urgent need for a fast‑acting, mechanistically broad neuroprotectant in TBI.
Invero has submitted an IND application for Severe Traumatic Brain Injury (sTBI) and is currently working with Vanderbilt University on a trial design. We are also pursuing Orphan Drug Designation for sTBI in intubated patients in the hospital setting.
Traumatic brain injury (TBI) is the leading cause of death and hallmark injury of U.S. military conflicts. A staggering half of a million U.S. service members suffered a TBI from 2000-2023. Among those patients hospitalized with TBI, one-third die and another one-half suffer long-term cognitive and functional impairment, which carries significant long-term morbidity. As a result, we are in communication with the Department of Defense to explore partnering opportunities to assess the positive effects of XENEX in sTBI patients.

Temporal Progression of Traumatic Brain Injury
This website uses cookies.
We use cookies to analyze website traffic and optimize your website experience. By accepting our use of cookies, your data will be aggregated with all other user data.
